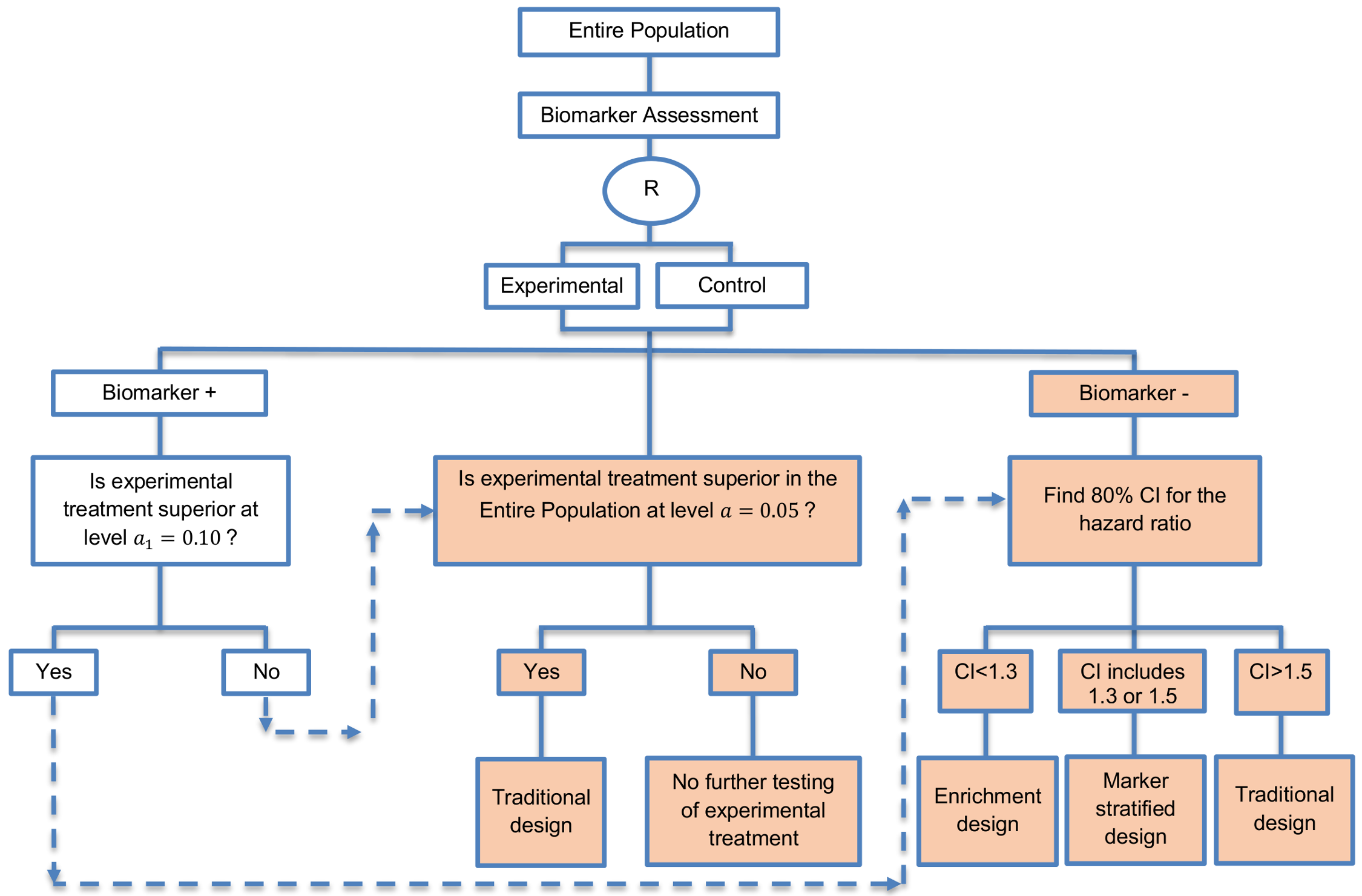
Freidlin et al., 2012 proposed a biomarker-guided Phase II clinical trial design in which when it is completed, it recommends which type of Phase III trial should be used. These recommendations for a Phase III trial are the following: (i) enrichment design; (ii) marker-stratified design; (iii) a traditional trial design without a biomarker; or (iv) drop consideration of the experimental treatment.
Alternative names: None Found
Details
Utility
Recommended when we want to conduct a Phase II randomized trial which allows decisions to be made about which type of Phase III biomarker-guided trial should be used.
Methodology
- Freidlin et al., 2012 [Google Scholar] have provided an online tool for calculating the sample size which can be found on the following website http://brb.nci.nih.gov/Data/FreidlinB/RP2BM.
- In order for a sample size to be estimated, the following information is required: (i) the significance levels for testing the treatment effect in the biomarker-positive subgroup and in the entire population; (ii) cut-offs and confidence intervals for the hazard ratio in the biomarker-negative subgroup; (iii) the prevalence of biomarker-positive patients; (iv) the median progression-free survival in each treatment arm in each biomarker-defined subgroup and (v) the accrual parameters.
- Regarding the accrual parameters, the author specifies the minimum sample size for biomarker-positive patients for which the accrual continues until this number is reached, the maximum number of over-accrual in biomarker-positive subgroup for which the accrual to the entire population stops after this number is reached and the maximum accrual number in biomarker-negative patients for which the accrual to this biomarker-defined subgroup stops when this number is reached.
Sample size Formula
Online tool for sample size calculation is available on the following website: http://brb.nci.nih.gov/Data/FreidlinB/RP2BM.
Statistical/Practical considerations
Advantages
- Works well in providing recommendations for phase III trial design.
Limitations
- None found
Key references
- Freidlin, B.; McShane, L.M.; Polley, M.-Y.C.; Korn, E.L. Randomized phase II trial designs with biomarkers. J. Clin. Oncol. 2012, 30, 3304–3309. [Google Scholar] [CrossRef] [PubMed]
Variations
No variations found for this trial design