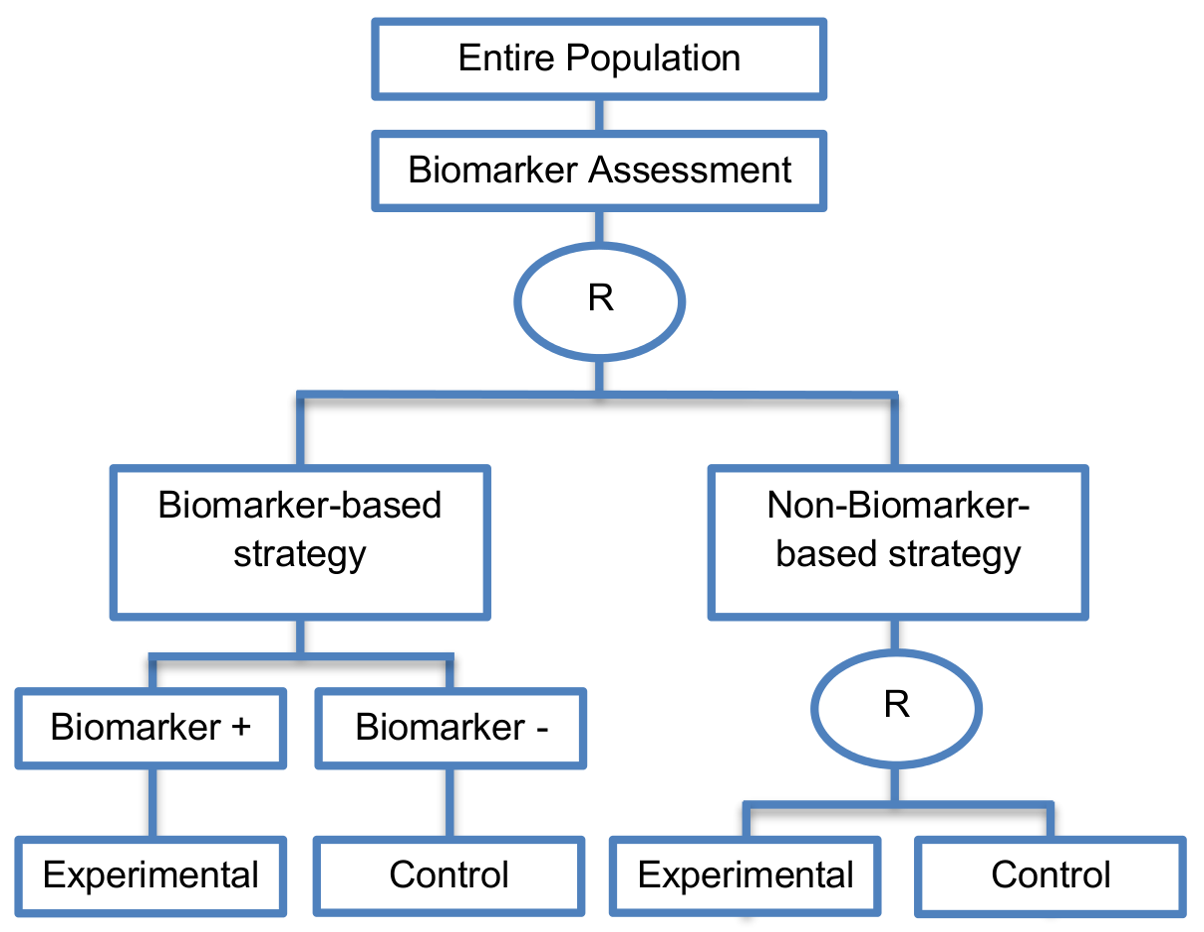
It includes a second randomization between experimental and control treatment in the non-biomarker guided strategy arm.
Alternative names: Biomarker-strategy designs with a randomized control, Modified marker-based strategy designs (for predictive biomarkers), Biomarker-strategy designs with randomized control, Marker-based designs with randomization in the non-marker-based arm, Marker-based strategy designs II, Marker-strategy designs, Augmented strategy designs, Trial designs allowing the evaluation of both the treatment and the marker effect
Details
Utility
In cases where we want to know whether the biomarker is not only prognostic but also predictive, these designs are preferable as compared to the Biomarker-Strategy Designs with Biomarker Assessment in the Control Arm and the Biomarker-Strategy Designs without Biomarker Assessment in the Control Arm.
Methodology
- The clinical utility of the biomarker is evaluated by comparing treatment effect between the biomarker-based strategy arm and non-biomarker-based strategy arm.
Sample size Formula
-
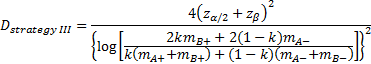
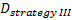 is referred to the required
total number of events (time-to-event outcome),
is referred to the required
total number of events (time-to-event outcome),
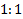 ratio between the two
treatment arms (experimental:control) is assumed,
ratio between the two
treatment arms (experimental:control) is assumed,
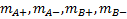 , denote the median survival
for biomarker-positive and biomarker-negative patients receiving control and
experimental treatments respectively.
, denote the median survival
for biomarker-positive and biomarker-negative patients receiving control and
experimental treatments respectively.
-
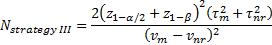
 is referred to the required
total sample size (continuous clinical endpoints),
is referred to the required
total sample size (continuous clinical endpoints),
 ratio between the two
treatment arms (experimental:control) is assumed,
ratio between the two
treatment arms (experimental:control) is assumed,
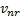 denotes the mean response from
the non-biomarker-based strategy arm,
denotes the mean response from
the non-biomarker-based strategy arm,
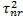 denotes the variance of
response for the non-biomarker-based strategy arm respectively.
denotes the variance of
response for the non-biomarker-based strategy arm respectively.
-
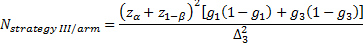
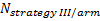 is referred to the required
total number of patients per arm (binary outcome),
is referred to the required
total number of patients per arm (binary outcome),
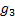 is the expected response rate
in the non biomarker-based strategy arm and
is the expected response rate
in the non biomarker-based strategy arm and
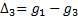 , the expected response rate
, the expected response rate
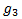 can be found by calculating the
formula
can be found by calculating the
formula
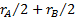 ,
,
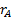 denotes the marginal effect of
treatment A (experimental treatment).
denotes the marginal effect of
treatment A (experimental treatment).
Statistical/Practical considerations
Advantages
- These designs have the ability to inform researchers about the potential superiority of the control treatment in the whole population or among a particular biomarker-defined subpopulation.
- Able to inform us whether the biomarker is prognostic or predictive.
- Allow clarification of whether the results which indicate efficacy of the biomarker-directed approach to treatment are caused due to a true effect of the biomarker status or to an improved treatment irrespective of the biomarker status.
- Galanis et al., 2011 [Google Scholar stated that these designs can be attractive when evaluating multiple biomarkers or the predictive value of molecular profiling between several treatment options is to be assessed. Also, Freidlin and Korn, 2010 [Google Scholar] claimed that these biomarker-strategy designs should be used only if researchers want to evaluate a complex biomarker-guided strategy with a variety of treatment options or biomarker categories.
Limitations
- Generally require a larger sample size as compared to the marker-stratified designs.
- Biomarker-positive and biomarker-negative subpopulations might be more imbalanced as compared with the first type of biomarker-strategy designs due to the fact that the randomization to different treatment strategies is performed before the evaluation of the biomarker status (balancing the randomization is useful to ensure that all randomized patients have tissue available). This can happen especially when the number of patients is very small.
Key references
- Gosho, M.; Nagashima, K.; Sato, Y. Study designs and statistical analyses for biomarker research. Sensors 2012, 12, 8966–8986. [Google Scholar] [CrossRef] [PubMed]
- Buyse, M. Towards validation of statistically reliable biomarkers. Eur. J. Cancer Suppl. 2007, 5, 89–95. [Google Scholar] [CrossRef]
- Young, K.Y.; Laird, A.; Zhou, X.H. The efficiency of clinical trial designs for predictive biomarker validation. Clin. Trials 2010, 7, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.J.; Xuemin, G.; Suyu, L. Bayesian adaptive randomization designs for targeted agent development. Clin. Trials 2010, 7, 584–596. [Google Scholar] [CrossRef] [PubMed]
- Hoering, A.; Leblanc, M.; Crowley, J.J. Randomized phase III clinical trial designs for targeted agents. Clin. Cancer Res. 2008, 14, 4358–4367. [Google Scholar] [CrossRef] [PubMed]
- Lader, E.W.; Cannon, C.P.; Ohman, E.M.; Newby, L.K.; Sulmasy, D.P.; Barst, R.J.; Fair, J.M.; Flather, M.; Freedman, J.E.; Frye, R.L.; et al. The clinician as investigator: Participating in clinical trials in the practice setting: Appendix 1: Fundamentals of study design. Circulation 2004, 109, e302–e304. [Google Scholar] [CrossRef] [PubMed]
- Galanis, E.; Wu, W.; Sarkaria, J.; Chang, S.M.; Colman, H.; Sargent, D.; Reardon, D.A. Incorporation of biomarker assessment in novel clinical trial designs: Personalizing brain tumor treatments. Curr. Oncol. Rep. 2011, 13, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.R.; Galanis, E. Incorporation of prognostic and predictive factors into glioma clinical trials. Curr. Oncol. Rep. 2013, 15, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Di Maio, M.; Gallo, C.; De Maio, E.; Morabito, A.; Piccirillo, M.C.; Gridelli, C.; Perrone, F. Methodological aspects of lung cancer clinical trials in the era of targeted agents. Lung Cancer 2010, 67, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Collette, L.; Bogaerts, J.; Suciu, S.; Fortpied, C.; Gorlia, T.; Coens, C.; Mauer, M.; Hasan, B.; Collette, S.; Ouali, M.; et al. Statistical methodology for personalized medicine: New developments at EORTC headquarters since the turn of the 21st century. Eur. J. Cancer Suppl. 2012, 10, 13. [Google Scholar] [CrossRef]
- Eickhoff, J.C.; Kim, K.; Beach, J.; Kolesar, J.M.; Gee, J.R. A bayesian adaptive design with biomarkers for targeted therapies. Clin. Trials 2010, 7, 546–556. [Google Scholar] [CrossRef] [PubMed]
- Ondra, T.; Dmitrienko, A.; Friede, T.; Graf, A.; Miller, F.; Stallard, N.; Posch, M. Methods for identification and confirmation of targeted subgroups in clinical trials: A systematic review. J. Biopharm. Stat. 2016, 26, 99–119. [Google Scholar] [CrossRef] [PubMed]
- Eng, K.H. Randomized reverse marker strategy design for prospective biomarker validation. Stat. Med. 2014, 33, 3089–3099. [Google Scholar] [CrossRef] [PubMed]
- Baker, S.G. Biomarker evaluation in randomized trials: Addressing different research questions. Stat. Med. 2014, 33, 4139–4140. [Google Scholar] [CrossRef] [PubMed]
- Mandrekar, S.J.; Grothey, A.; Goetz, M.P.; Sargent, D.J. Clinical trial designs for prospective validation of biomarkers. Am. J. Pharmacogenom. 2005, 5, 317–325. [Google Scholar] [CrossRef]
- Therasse, P.; Carbonnelle, S.; Bogaerts, J. Clinical trials design and treatment tailoring: General principles applied to breast cancer research. Crit. Rev. Oncol. Hematol. 2006, 59, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Sargent, D.; Allegra, C. Issues in clinical trial design for tumor marker studies. Semin. Oncol. 2002, 29, 222–230. [Google Scholar] [CrossRef] [PubMed]